This post is part of a series on graphical metaphors (continue here).
There are many sequences that have been proposed to be suited for layer-fMRI. This list includes GE-BOLD (Menon 1999), SE-EPI (Goense 2006), CPMG (Scheffler 2021), ASL (Kashyap 2021), diffusion fMRI (Truong 2009), 3D-GRASE (Moerel 2018), calibrated BOLD aka CMRO2 mapping (Guidi 2020), VASO (Hua 2013), phase regression (Stanley 2021), onset-time imaging (Yu 2014), depth-dependent deconvolution (Markuerkiaga 2021), CVR-calibration (Guidi 2016), and many more.
In this blog post, I want to summarise the take-home message from the seemingly never ending battle between researchers fighting about the best sequence for layer-fMRI. I seek to do so by means of cynical metaphors in graphical form. Because, why not. There are plenty more serious discussions already elsewhere1,2,3.
I counted several dozen published studies that aim to establish a new sequence for layer-fMRI and compare it with other more conventional methods to convince the field about its superiority.

Many resources of the field have been invested.

There is a common consensus that is somewhat agreed upon between most researchers.

Since many experimenters consider the detection sensitivity as the single most limiting factor of doing a layer-fMRI study, GE-BOLD is by far the most popular method to date.
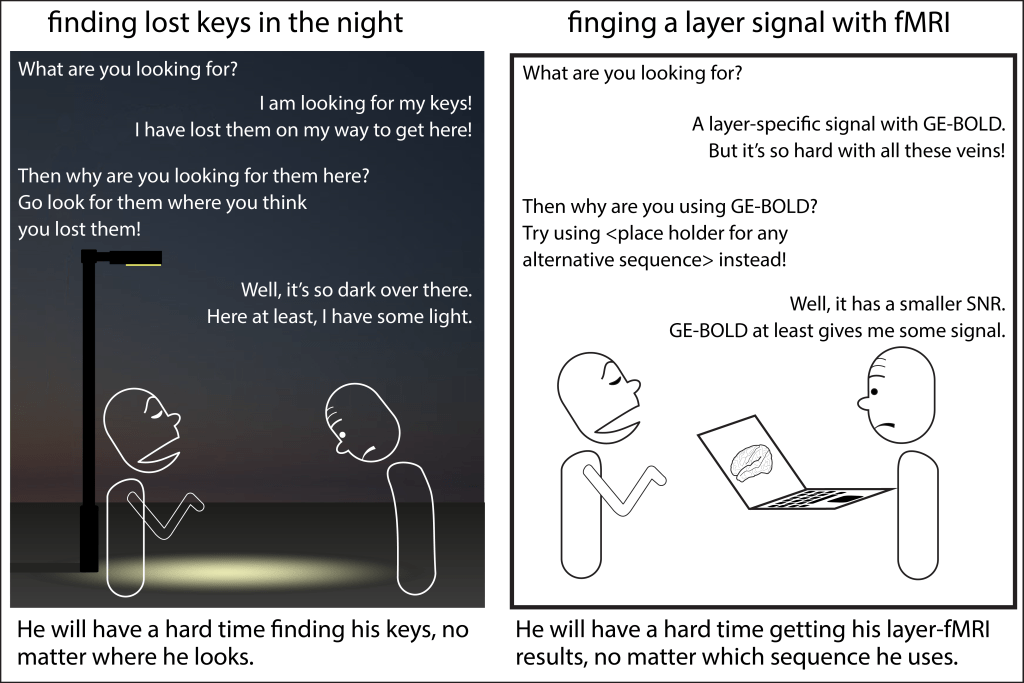
The question of the best layer-fMRI sequence is extremely popular in the field. And also I – myself have contributed my fair share of comparison studies to the literature. Was it well invested mental energy to search for the best sequence?

And even within very specific benchmarking test cases, there is rarely an ultimatum falsifiable conclusion.

Non-quantitative non-scientific aspects are important too. E.g. the sequence’s availability of your vendor, and your personal experience (skill) of a given sequence can be more important than the aspects that are discussed in the academic literature (sensitivity and specificity).
The fact that sequences are made by researchers and are also simultaneously seen as their object of research, results in weird studies.


Since different sequences perform differently well in different experimental setups, we can look forward to many decades of future comparison studies.

One potential solution out of this dilemma is to refrain from future comparison studies and rather shift the focus on optimising the experimental setup of the experiment.

Alternatively, we could also continue with the endless series of comparison studies and just make the most of it. E.g. We could start measuring our success not based on the best sequence but rather on all the other new technologies that are being developed along the way.

Or, we just play it safe and are a bit more wasteful about our scan time resourced. Neuroscientists are so generous about scan time and the number of control conditions. Let’s be more like them.

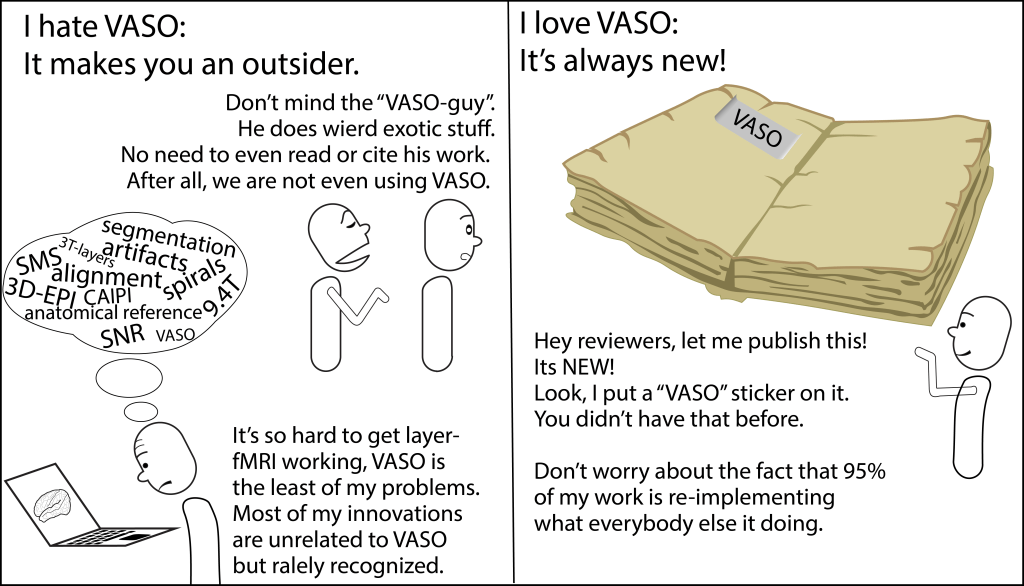
And there are certainly some career implications of working with unconventional sequences.
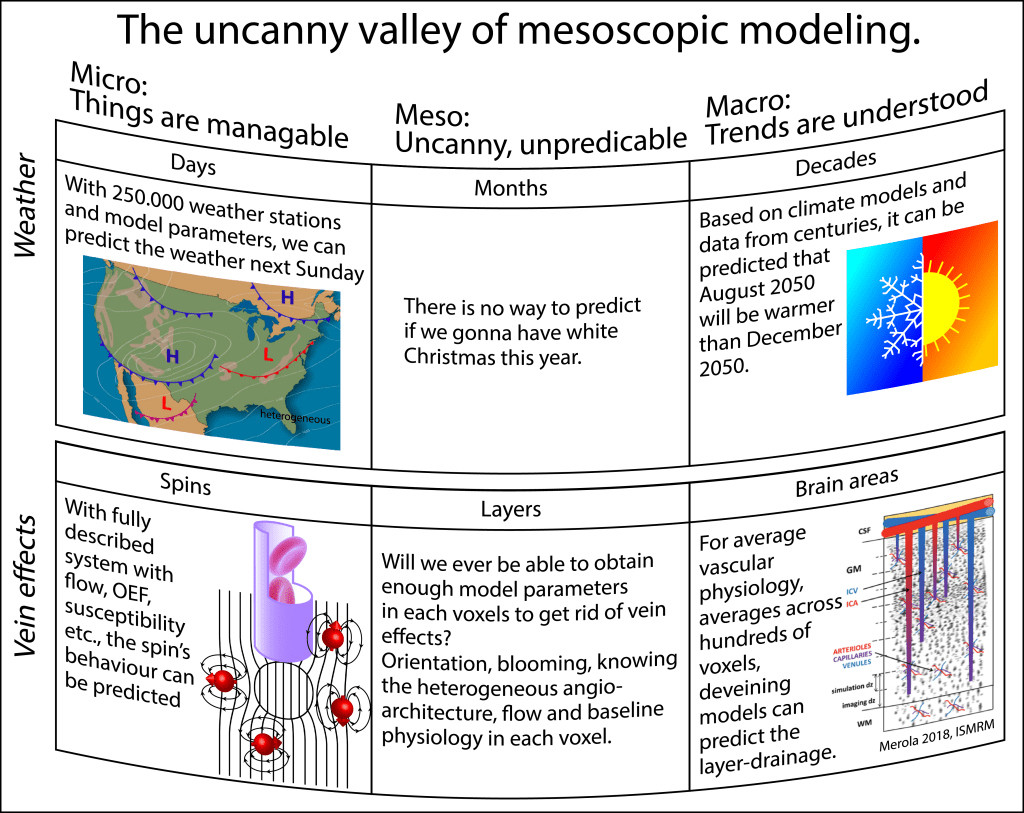
Can’t we just model it and solve all the sequence problems on the analysis side?

Acknowledgements
This post is based on the 1st virtual layer-fMRI dinner 2020. A condensed version of this blog post is provided to Luca Vizioli and Essa Yacoub to be considered as part of our book chapter “laminar imaging at UHF” in the book “Ultra-high field neuro MRI” edited by Maxime Guye, Karin Markenroth Bloch and Benedikt A. Poser. Please note that metaphors are figurative means to evoke a certain effect without fully resembling the complexity of the real world.
References
- Guidi M, Huber L, Lampe L, Merola A, Ihle K, Möller HE. Cortical laminar resting-state fluctuations scale with the hypercapnic bold response. HBM. 2020;41:2014-2027. doi:10.1002/hbm.24926
- Guidi M, Huber L, Lampe L, Gauthier CJ, Möller HE. Lamina-dependent calibrated BOLD response in human primary motor cortex. Neuroimage. 2016;141:250-261. doi:10.1016/j.neuroimage.2016.06.030
- Haenelt D, Weiskopf N, Vaculciakova L, et al. Mapping Ocular Dominance Columns in Humans Using GE-EPI, SE-EPI and SS-SI-VASO at 7 T. In: Proc Intl Soc Mag Reson Med. ; 2020:1230. https://cds.ismrm.org/protected/20MPresentations/abstracts/1230.html.
- Hua J, Jones CK, Qin Q, Van Zijl PCM. Implementation of vascular-space-occupancy MRI at 7T. Magn Reson Med. 2013;69(4):1003-1013. doi:10.1002/mrm.24334
- Huber L (Renzo). Whatever works: There is no winner among BOLD or non-BOLD contrasts. Proc first layer-fMRI Dinn. 2020. doi:10.5281/zenodo.3874364
- Kashyap S, Ivanov D, Havlicek M, Huber L, Poser BA, Uludağ K. Sub-millimetre resolution laminar fMRI using Arterial Spin Labelling in humans at 7 T. PLoS One. 2021;16(4 April):1-23. doi:10.1371/journal.pone.0250504
- Markuerkiaga I, Marques P, Gallagher TE, Norris DG. Estimation of laminar BOLD activation profiles using deconvolution with a physiological point spread function. J Neurosci Methods. 2021;353:109095. doi:10.1016/j.jneumeth.2021.109095
- Menon RS, Goodyear BG. Submillimeter functional localization in human striate cortex using BOLD contrast at 4 Tesla: Implications for the vascular point-spread function. Magn Reson Med. 1999;41(2):230-235. doi:10.1002/(SICI)1522-2594(199902)41:2<230::AID-MRM3>3.0.CO;2-O
- Merola A, Weiskopf N. Modelling the laminar GRE-BOLD signal: integrating anatomical, physiological and methodological determinants. In: Proc Intl Soc Mag Reson Med. ; 2018:2299. https://cds.ismrm.org/protected/18MPresentations/abstracts/2299.html.
- Moerel M, De Martino F, Kemper VG, et al. Sensitivity and specificity considerations for fMRI encoding, decoding, and mapping of auditory cortex at ultra-high field. Neuroimage. 2018;164(March):18-31. doi:10.1016/j.neuroimage.2017.03.063
- Olman C. Whatever gives me the best true resolution and decent SNR. In: Proceedings of the First Layer-FMRI Dinner. ; 2020:3. doi:10.5281/zenodo.3874364
- Polimeni JR, Fischl B, Greve DN, Wald LL. Laminar analysis of 7 T BOLD using an imposed spatial activation pattern in human V1. Neuroimage. 2010;52(4):1334-1346. doi:10.1016/j.neuroimage.2010.05.005
- Scheffler K, Engelmann J, Heule R. BOLD sensitivity and vessel size specificity along CPMG and GRASE echo trains. Magn Reson Imaging. 2021:1-8. doi:10.1002/mrm.28871
- Stanley OW, Kuurstra AB, Klassen LM, Menon RS, Gati JS. Effects of phase regression on high-resolution functional MRI of the primary visual cortex. Neuroimage. 2021;227(December 2020):117631. doi:10.1016/j.neuroimage.2020.117631
- Truong TK, Song AW. Cortical depth dependence and implications on the neuronal specificity of the functional apparent diffusion coefficient contrast. Neuroimage. 2009;47(1):65-68. doi:10.1016/j.neuroimage.2009.04.045
- Yu X, Qian C, Chen D-Y, Dodd SJ, Koretsky AP. Deciphering laminar-specific neural inputs with line-scanning fMRI. Nat Methods. 2014;11:55-58. doi:10.1038/nmeth.2730
One thought on “Which sequence is best for layer-fMRI? A graphic story in cynical metaphors.”