This post contains a collection of recently presented powerpoint slides.
Author: renzohuber
Documentation of Installing an IDEA VirtualBox for VE11 from OVA
This post documents the installation of an IDEA VE11 virtual box on a mac as done on May 14th 2018
Big thanks to Andy for figuring out how this works
Prerequisites
- Here I start with a already built images of IDEA on windows vista and mars on Ubuntu. the images from FMRIF can be taken from erbium.nimh.nih.gov:/fmrif/projects/SiemensIdea/virtual_machines/OVF/): IDEA_ve11c-mars.ova and IDEA_ve11c+vd13d+vd13a.ova
- Virtual box software can be downloaded here.
Continue reading “Documentation of Installing an IDEA VirtualBox for VE11 from OVA”
EPI phase correction algorithms
At high resolution EPI, the gradients are pushed to their limits and the ramp sampling ratio is particularly large. This means that the ghosting is increased and the Nyquist ghost correction is getting more important. In this post, I describe how to change the Nyquist ghost correction algorithm.

SS-SI VASO pitfalls in visual cortex

With respect to high-resolution VASO application, visual cortex is very unique. I found it to be a challenging area. However, because of its high demand, I have been working on is with multiple collaborators. The most important pitfalls of SS-SI VASO in visual cortex that I came across in these collaborations are discussed below.
The take home message tat I learned from manny experiments is:
- Use axial slices with the phase encoding direction A>>P.
- Watch out for negative voxels.
- Invest a lot of effort in optimizing GRAPPA parameters, its worth it.
Unwanted spatial blurring during resampling
In layer-fMRI, we spend so much time and effort to achieve high spatial resolutions and small voxel sizes during the acquisition. However, during the evaluation pipeline much of this spatial resolution can be lost during multiple resampling steps.
In this post, I want to discuss sources of signal blurring during spatial resampling steps and potential strategies to account for them.
Continue reading “Unwanted spatial blurring during resampling”
Partial-Fourier imaging at High Resolutions
This blog post discusses the resolution loss when applying partial-Fourier imaging in GE-EPI in the presence of strong T2*-decay.
I found that that when I was aiming for high-resolutions, it is beneficial to refrain from the application of partial Fourier (PF) imaging as much as possible. For the long readout durations at high-resolutions and the fast T2/T2*-decay at high field strengths results in even stronger blurring of partial-Fourier.
Continue reading “Partial-Fourier imaging at High Resolutions”
Smoothing within layers
Smoothing within layers can be advantageous for multiple reasons:
- Increasing the CNR without loosing spatial information across cortical depths.
- Visualization of striping pattern across columnar structures.
- Avoiding leakage of physiological noise from CSF space into GM tissue.
Finding ROI of the double layers in M1
In this post I want to describe the guidelines that helped me to find the right spot of primary motor cortex (M1) that has a double-layer pattern during a conventional finger tapping task.
The motor cortex is an excellent model system to debug-layer fMRI methodology for multiple reasons:
- It has a consistent folding pattern across people.
- Its folding pattern is convoluted across one axis only. Hence, it is possible to use thicker slices with higher in-plane resolution.
- With 4mm, its is the thickest part of the cortex compared to all other areas. Hence, layer analysis can be done even with 1.2 mm voxels.
- It has an expected double layer structure, with two separate peaks. The separability of the peaks can be used as a measure of functional specificity.
- It is very close to the RF-receive coils and has high tSNR.
- It is easy to shim.
One tricky part, however, is to find the right location of the double layer feature.
Quick analysis pipeline of getting layer fMRI profiles without anatomical reference data
This is a step-by-step description on how to obtain layer profiles from any high-resolution fMRI dataset. It is based on manual delineated ROIs and does not require the tricky analysis steps including distortion correction, registration to whole brain “anatomical” datasets, or automatic tissue type segmentation. Hence this is a very quick way for a first glance of the freshly acquired data.

The important steps are: 1.) Upscaling, 2.) Manual delineation of GM, 3.) Calculation of cortical depths in ROI, 4.) Extracting functional data based on calculated cortical depths.
Gradient Temperature
tSNR changes across time
I got interested in gradient temperature because of the weird effect that tSNR seemed to increase over time.
Upon posing this effect on Twitter, PracitalfMRI and Ben Poser suggested that it might be due to gradient temperature. So I learned how to track it with as described below.
Protected: Literature review of digit mapping in motor cortex
GRAPPA kernel size
Almost every modern fMRI protocol (at SIEMENS scanners) uses GRAPPA. However, only very few people pay a lot of attention on optimal usage of the GRAPPA auto-callibration data. I realized the importance of optimizing GRAPPA parameters when doing high-resolution EPI. At high resolutions, GRAPPA-related noise can become an increasingly important limitation. This is especially true with the low bandwidth that the body gradient coils force us to use.
In this blog-post I will explain how the GRAPPA kernel-size affects the fMRI data quality, how you can change it, how you can find out which kernel-size was used, and I will descrive simple software tools to identify regions that might benefit from adaptations of the GRAPPA-kernel size.
layer-fMRI software repositories
There is a long list of software packages that are capable of performing high-resolution MRI analysis.
Some of them are used by multiple groups and some of them are customized for specific studies only.
In this post, I want to give an overview over the most important software packages, their advantages and disadvantages, and their popularity in the field.
Layer-fMRI researcher
There are about 80 people with a strong interest in layer-fMRI.
The whole list can be seen here.
These people are included in the listserv: laminar.imaging.network@gmail.com
To subscribe, please write an informal email to laminar.imaging.network@gmail.com
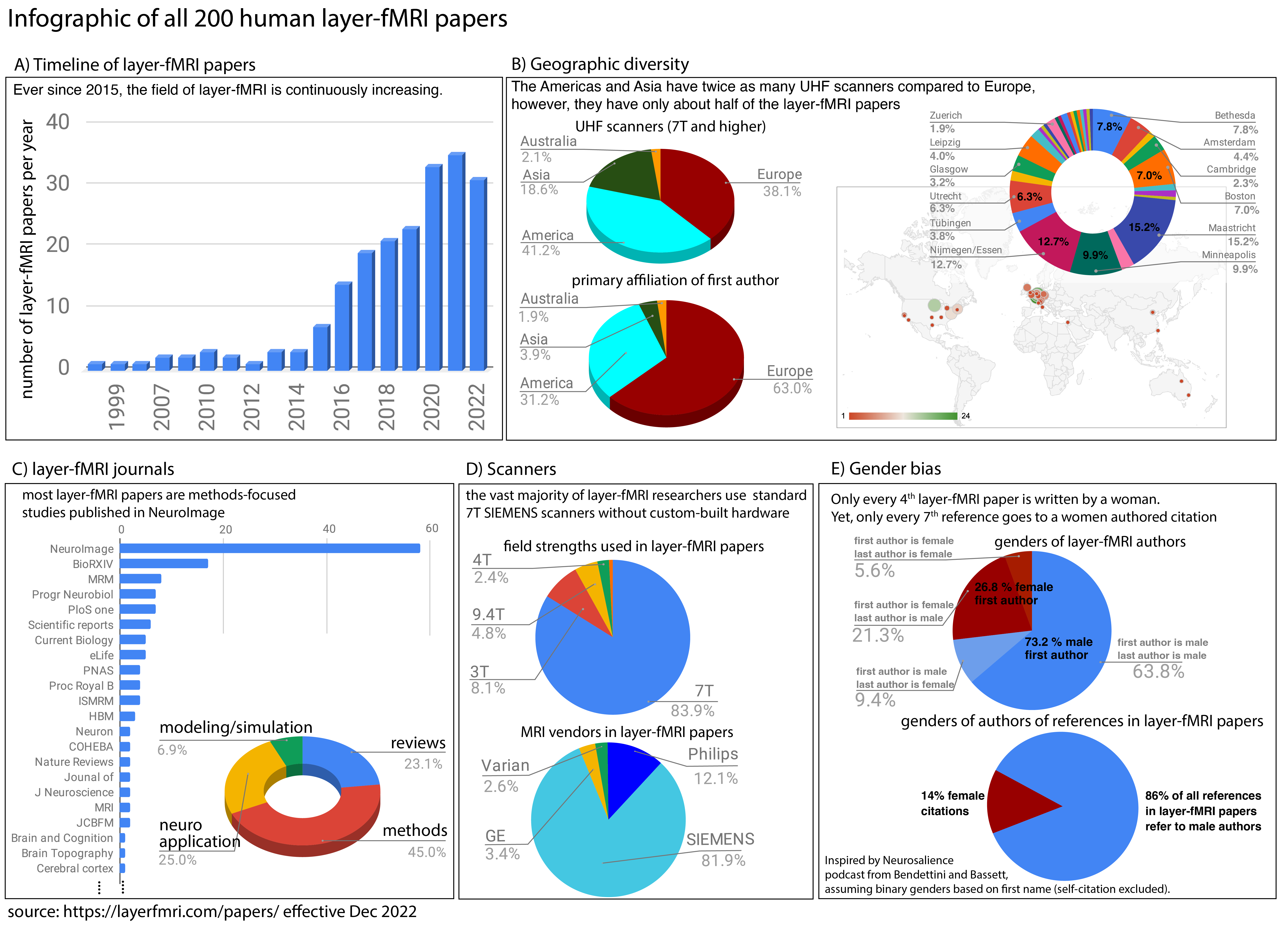
Layer fMRI papers
This page depicts a collection of all layer-fMRI papers.
This list solely includes papers that fulfill the following inclusion criteria:
- focus on functional imaging
- cortical layers (and/or sub-millimeter resolutions)
- human imaging
- preprints are included
Suggestions and corrections are welcome layerfMRI@gmail.com
For non-cortical, non-fMRI, non-human, or non-layer high resolution MRI, please see reviews in special issues 1, 2, 3 (and references therein). The raw data used in this infographic are available here.
Before and after COVID effects.

Click on image to enlarge.
2025
List is discontinued in April 2025. There are just too many papers. I am continuing to maintain the list on Gdrive. With the ambition to be comprehensive here.
- Chai, Y., Li, L., Stirnberg, R., Huber, L., Stöcker, T., Bandettini, P.A., Sutton, B.P., 2025. Blood-nulling versus tissue-suppression: Enhancing integrated VASO and perfusion (VAPER) contrast for laminar fMRI. Imaging Neuroscience.
- Faes, L.K., Zulfiqar, I., Vizioli, L., Yu, Z., Wu, Y.-H., Shin, J., Cloos, M.A., Auksztulewicz, R., Melloni, L., Uludag, K., Yacoub, E., De Martino, F., 2025. Predictive acoustical processing in human cortical layers. bioRxiv.
- Knudsen, L., Jespersen, B., Heintzelmann, M.B., Zhang, P., Yang, Y., Lund, T.E., Blicher, J.U., 2024. Laminar fMRI in the locked-in stage of amyotrophic lateral sclerosis shows preserved activity in layer Vb of primary motor cortex. Frontiers in Human Neuroscience.
2024
- Huber, R., Stirnberg, R., Morgan, A.T., Feinberg, D.A., Ehses, P., Gulban, O.F., Koiso, K., Swegle, S., Gephart, I., Wardle, G., Persichetti, A., Beckett, A.J., Stöcker, T., Boulant, N., Poser, B.A., Bandettini, P., 2024. Fuzzy ripple artifact in high resolution fMRI: identification, cause, and mitigation. bioRxiv.
- Straub, S., Zhou, X., Tao, S., Westerhold, E.M., Jin, J., Middlebrooks, E.H., 2024. Feasibility of laminar functional quantitative susceptibility mapping. bioRxiv.
- Kan, C. (Kenny), Stirnberg, R., Montequin, M., Gulban, O.F., Morgan, A.T., Bandettini, P., Huber, L., 2024. T1234: A distortion-matched structural scan solution to misregistration of high resolution fMRI data.
- Degutis, J.K., Chaimow, D., Haenelt, D., Assem, M., Duncan, J., Haynes, J.-D., Weiskopf, N., Lorenz, R., 2024. Dynamic layer-specific processing in the prefrontal cortex during working memory. Commun Biol 7, 1140.
- Chai, Y., Morgan, A.T., Handwerker, D.A., Li, L., Huber, L., Sutton, B.P., Bandettini, P.A., 2024. Improving laminar fMRI specificity by reducing macrovascular bias revealed by respiration effects. Imaging Neuroscience.
- Pfaffenrot, Bouyeure, Gomes, Kashyap, Axmacher, Norris. 2024 Characterizing BOLD activation patterns in the human hippocampus with laminar fMRI. BioRxiv.
- Thomas, E.R., Haarsma, J., Nicholson, J., Yon, D., Kok, P., Press, C., 2024. Predictions and errors are distinctly represented across V1 layers. Current Biology 34, 2265-2271.e4.
- Sterzer, P., Keller, G.B., 2024. Predictive processing: Layer-specific prediction error signals in human cortex. Current Biology 34, R496–R498.
- Shamir, I., Assaf, Y., 2024. Laminar RNNs: using biologically-inspired network topology on the cortical laminar level in memory tasks.
- Knudsen, L., Jespersen, B., Heintzelmann, M.B., Zhang, P., Yang, Y., Lund, T.E., Blicher, J.U., 2024. Laminar fMRI in the locked-in stage of amyotrophic lateral sclerosis shows preserved activity in layer Vb of primary motor cortex.
- Warrington, O., Graedel, N.N., Callaghan, M.F., Kok, P., n.d. Communication of perceptual predictions from the hippocampus to the deep layers of the parahippocampal cortex 2024, BioRxiv.
- Wang, Y., Qian, C., Gao, Y., Zhou, Y., Zhang, X., Wen, W., Zhang, P., 2024. Amblyopic deficits in monocular processing and binocular interactions revealed by submillimeter 7T fMRI and EEG frequency tagging.
- Chai, Y., Morgan, A.T., Xie, H., Li, L., Huber, L., Bandettini, P.A., Sutton, B.P., 2024. Unlocking near-whole brain, layer-specific functional connectivity with 3D VAPER fMRI. Imaging Neuroscience.
- Iyyappan Valsala, P., Veldmann, M., Bosch, D., Scheffler, K., Ehses, P., 2024. Submillimeter balanced SSFP BOLD–functional MRI accelerated with 3D stack‐of‐spirals at 9.4 T. Magnetic Resonance in Med mrm.30064.
- Dresbach, S., Huber, R., Gulban, O.F., Pizzuti, A., Trampel, R., Ivanov, D., Weiskopf, N., Goebel, R., 2024. Characterisation of laminar and vascular spatiotemporal dynamics of CBV and BOLD signals using VASO and ME-GRE at 7T in humans. bioRxiv.
- Nasr, S., Skerswetat, J., Gaier, E.D., Malladi, S.N., Kennedy, B., Tootell, R.B.H., Bex, P., Hunter, D.G., 2024. Using high-resolution functional MRI to differentiate impacts of strabismic and anisometropic amblyopia on evoked ocular dominance activity in humans (preprint). Neuroscience.
2023
- Huber, L., Kronbichler, L., Stirnberg, R., Ehses, P., Stöcker, T., Fernández-Cabello, S., Poser, B.A., Kronbichler, M., 2023. Evaluating the capabilities and challenges of layer-fMRI VASO at 3T 3.
- Haarsma, J., Deveci, N., Corbin, N., Callaghan, M.F., Kok, P., 2023. Expectation cues and false percepts generate stimulus-specific activity in distinct layers of the early visual cortex Laminar profile of visual false percepts. J. Neurosci. JN-RM-0998-23.
- Degutis, J.K., Chaimow, D., Haenelt, D., Assem, M., Duncan, J., Haynes, J.-D., Weiskopf, N., Lorenz, R., 2023. Dynamic layer-specific processing in the prefrontal cortex during working memory (preprint). Neuroscience.
- Chang, W.-T., Lin, W., Giovanello, K.S., 2023. Enabling brain-wide mapping of directed functional connectivity at 3T via layer-dependent fMRI with draining-vein suppression (preprint). Neuroscience.
- Faes, L.K., De Martino, F., Huber, L. (Renzo), 2023. Cerebral blood volume sensitive layer-fMRI in the human auditory cortex at 7T: Challenges and capabilities. PLoS ONE 18, e0280855.
- Haenelt, D., Chaimow, D., Nasr, S., Weiskopf, N., Trampel, R., 2023. Decoding of columnar-level organization across cortical depth using BOLD- and CBV-fMRI at 7 T. bioRxiv.
- Yun, S.D., Küppers, F., Shah, N.J., 2023. Submillimeter fMRI Acquisition Techniques for Detection of Laminar and Columnar Level Brain Activation. Magnetic Resonance Imaging jmri.28911.
- Logan T. Dowdle, Luca Vizioli, Steen Moeller, Mehmet Akçakaya, Cheryl Olman, Geoffrey Ghose, Essa Yacoub, Kâmil Uğurbil., 2023, Evaluating Increases in Sensitivity from NORDIC for Diverse fMRI Acquisition Strategies. NeuroImage,
- Heij, J., Raimondo, L., Siero, J.C.W., Dumoulin, S.O., Van Der Zwaag, W., Knapen, T., 2023. A selection and targeting framework of cortical locations for line‐scanning fMRI. Human Brain Mapping hbm.26459.
- Báez‐Yáñez, M.G., Siero, J.C.W., Petridou, N., 2023. A mechanistic computational framework to investigate the hemodynamic fingerprint of the blood oxygenation level‐dependent signal. NMR in Biomedicine e5026.
- Malekian, V., Graedel, N.N., Hickling, A., Aghaeifar, A., Dymerska, B., Corbin, N., Josephs, O., Maguire, E.A., Callaghan, M.F., 2023. Mitigating susceptibility-induced distortions in high-resolution 3DEPI fMRI at 7T. NeuroImage 120294.
- Qian, C., Chen, Z., de Hollander, G., Knapen, T., Zhang, Z., He, S., Zhang, P., 2023. Hierarchical cortical and subcortical mechanisms underlying binocular rivalry (preprint). Neuroscience.
- Demirel, Ö.B., Moeller, S., Vizioli, L., Yaman, B., Dowdle, L., Yacoub, E., Uğurbil, K., Akçakaya, M., 2023. High-Quality 0.5mm Isotropic fMRI: Random Matrix Theory Meets Physics-Driven Deep Learning, IEEE,, MD, USA, pp. 1–6.
2022
- de Oliveira ÍAF, Siero JCW, Dumoulin SO, van der Zwaag W. Improved Selectivity in 7 T Digit Mapping Using VASO-CBV. Brain Topogr [Internet]. 2022 Dec 14 [cited 2022 Dec 19]
- Dowdle LT, Ghose G, Moeller S, Ugurbil K, Yacoub E, Vizioli L. Task Demands Differentiate Regional Depth-Dependent Activity Profiles Within the Ventral Visual Pathway. 2022 BioRxiv
- Pfaffenrot V, Koopmans PJ. Magnetization Transfer weighted laminar fMRI with multi-echo FLASH. NeuroImage. 2022 Oct;119725.
- Demirayak P, Deshpande G, Visscher K. Laminar functional magnetic resonance imaging in vision research. Front Neurosci. 2022 Oct 4;16:910443.
- Deshpande G, Wang Y. Noninvasive Characterization of Functional Pathways in Layer-Specific Microcircuits of the Human Brain Using 7T fMRI. Brain Sciences. 2022 Oct 7;12(10):1361.
- Han S, Eun S, Cho H, Uludaǧ K, Kim SG. Improved laminar specificity and sensitivity by combining SE and GE BOLD signals. NeuroImage. 2022 Oct;119675.
- Merriam EP, Gulban OF, Kay K. The need for validation in layer-specific fMRI [Internet]. Open Science Framework; 2022.
- Pizzuti, Alessandra, Laurentius (Renzo) Huber, Omer Faruk Gulban, Amaia Benitez-Andonegui, Judith Peters, and Rainer Goebel. “Imaging the Columnar Functional Organization of Human Area MT+ to Axis-of-Motion Stimuli Using VASO at 7 Tesla.” Preprint. Neuroscience, August 1, 2022.
- Koiso, Kenshu, Anna K Müller, Kazuaki Akamatsu, Sebastian Dresbach, Omer Faruk Gulban, Rainer Goebel, Yoichi Miyawaki, Benedikt A Poser, and Laurentius Huber. “Acquisition and Processing Methods of Whole-Brain Layer-FMRI VASO and BOLD: The Kenshu Dataset.” .
- Huber, Laurentius (Renzo), Panagiotis Kassavetis, Omer Faruk Gulban, Mark Hallett, and Silvina G Horovitz. “Laminar VASO FMRI in Focal Hand Dystonia Patients.” Preprint. Neuroscience, August 1, 2022.
- Faes, Lonike K., Federico De Martino, and Laurentius (Renzo) Huber. “Cerebral Blood Volume Sensitive Layer-FMRI in the Human Auditory Cortex at 7 Tesla: Challenges and Capabilities.” Preprint. Neuroscience, August 3, 2022.
- Huber, L., Kronbichler, L., Stirnberg, R., Poser, B. A., Fernández-Cabello, S., Stöcker, T., & Kronbichler, M. (2021). Evaluating the capabilities and challenges of layer-fMRI VASO at 3T. BioRxiv, 1229.
- Liu, T., Fu, J., Japee, S., Chai, Y., Ungerleider, L., & Merriam, E. (2020). Layer-specific modulation of visual responses in human visual cortex by emotional faces. Journal of Vision, 20(11), 587.
- Graedel, N. N., Miller, K. L., & Chiew, M. (2022). Ultrahigh Resolution fMRI at 7T Using Radial-Cartesian TURBINE Sampling. Magn Res Med, May, 1–16.
- Chang, W., Langella, S., & Giovanello, K. (2022). Cross-layer Balance of Visuo-hippocampal Functional Connectivity Is Associated With Episodic Memory Recognition Accuracy. Research Square, 1–21.
- Haarsma, J., Deveci, N., Corbin, N., Callaghan, M., & Kok, P. (2022). Perceptual expectations and false percepts generate stimulus-specific activity in distinct layers of the early visual cortex. BioRxiv, 1–12.
- Knudsen, L., Bailey, C. J., Blicher, J. U., Yang, Y., Zhang, P., & Torben, E. (2022). Feasibility of 3T layer-dependent fMRI with GE-BOLD using NORDIC and phase regression. BioRxiv, 1–26.
- Scheeringa, R., Bonnefond, M., Van Mourik, T., Jensen, O., Norris, D. G., & Koopmans, P. J. (2020). Relating neural oscillations to laminar fMRI connectivity. BioRxiv, 2020.09.18.303263.
- Yun SD, Pais-roldán P, Palomero-gallagher N, Shah NJ. Mapping of Whole-Brain Resting-State Networks with Half-Millimetre. HBM. 2022.
- Deshpande, G., Zhao, Robinson, J. (2022). Functional Parcellation of the Hippocampus based on its Layer-specific Connectivity with Default Mode and Dorsal Attention Networks. NeuroImage, 119078.
- Cerliani L, Bhandari R, De Angelis L, et al. Predictive coding during action observation – a depth-resolved intersubject functional correlation study at 7T. Cortex. 2022.
- Deshpande G, Wang Y, Robinson J. Resting state fMRI connectivity is sensitive to laminar connectional architecture in the human brain. Brain Informatics. 2022;9(1).
- Iamshchinina P, Haenelt D, Trampel R, Weiskopf N, Kaiser D, Cichy RM. Benchmarking GE-BOLD, SE-BOLD, and SS-SI-VASO sequences for depth-dependent separation of feedforward and feedback signals in high-field MRI. bioRxiv. 2022:1-18.
- Yu Y, Huber L, Yang J, et al. Layer-specific activation in human primary somatosensory cortex during tactile temporal prediction error processing. Neuroimage. 2022;248:118867.
- Kurzawski JW, Gulban OF, Jamison K, Winawer J, Kay KN. The influence of non-neural factors on BOLD signal magnitude. bioRxiv. 2021;1822683:1-25.
- Akbari A, Bollmann S, Ali TS, Barth M. Modelling the depth-dependent VASO and BOLD responses in human primary visual cortex. HBM. 2022:1-31.
2021
- Molaei-Vaneghi, Fatemeh, Natalia Zaretskaya, Tim van Mourik, Jonas Bause, Klaus Scheffler, and Andreas Bartels. “Integration of Visual Motion and Pursuit Signals in Areas V3A and V6+ across Cortical Depth Using 9.4T FMRI.” Preprint. Neuroscience, December 10, 2021. .
- Vizioli L, Yacoub E, Lewis LD, How pushing the spatiotemporal resolution of fMRI can advance neuroscience. Progress in Neurobiology, 2021.
- Schellekens W, Bhogal AA, Roefs ECA, Báez-Yáñez MG, Siero JCW, Petridou N. The many layers of BOLD. On the contribution of different vascular compartments to laminar fMRI. bioRxiv. 2021:6.
- Ng AKT, Jia K, Goncalves NR, et al. Ultra-High-Field Neuroimaging Reveals Fine-Scale Processing for 3D Perception. J Neurosci. 2021;41(40):8362-8374.
- Zwart JA De, Gelderen P Van, Duyn JH. Sensitivity limitations of high-resolution perfusion-based human fMRI at 7 Tesla. Magn Reson Imaging. 2021;84:135-144.
- Zoraghi M, Scherf N, Jaeger C, et al. Simulating Local Deformations in the Human Cortex Due to Blood Flow-Induced Changes in Mechanical Tissue Properties: Impact on Functional Magnetic Resonance Imaging. Front Neurosci. 2021;15(September):1-13.
- Iamshchinina P, Kaiser D, Yakupov R, et al. Perceived and mentally rotated contents are differentially represented in cortical depth of V1. Commun Biol. 2021:1-8.
- Pfaffenrot V, Voelker MN, Kashyap S, Koopmans PJ. Laminar fMRI using T2-prepared multi-echo FLASH. Neuroimage 2021;236(236):118163
- Cerliani L, Bhandari R, Angelis L De, et al. Depth-resolved intersubject functional correlation of 7T BOLD signals reveals increased stimulus related information sharing across deep layers in premotor and parietal nodes for predictable actions. bioRxiv. 2021:1-33.
- Vizioli L, Moeller S, Dowdle L, et al. Lowering the thermal noise barrier in functional brain mapping with magnetic resonance imaging. Nat Commun. 2021;12:5181.
- Ng AKT, Jia K, Goncalves NR, et al. Ultra-high field neuroimaging reveals fine-scale processing for 3D perception. J Neurosci. 2021;JN-RM-0065(July). doi:10.1523/JNEUROSCI.0065-21.2021
- Raimondo L, Knapen T, Oliveira ĺcaro A., et al. A line through the brain: implementation of human line-scanning at 7T for ultra-high spatiotemporal resolution fMRI. J Cereb Blood Flow Metab. 2021:0271678X2110372.
- van Dijk JA, Fracasso A, Petridou N, Dumoulin SO. Laminar processing of numerosity supports a canonical cortical microcircuit in human parietal cortex. Curr Biol. 2021:1-6.
- Chai Y, Liu TT, Marrett S, et al. Topographical and laminar distribution of audiovisual processing within human planum temporale. Prog Neurobiol. 2021;(July):102121.
- Chai Y, Li L, Wang Y, et al. Magnetization Transfer Weighted EPI Facilitates Cortical Depth Determination in Native fMRI Space. Neuroimage. 2021:118455.
- Han S, Eun S, Cho H, Uluda K, Kim S. Improvement of sensitivity and specificity for laminar BOLD fMRI with double spin-echo EPI in humans at 7 T. Neuroimage. 2021;241(241):118435.
- Bandettini PA, Huber L, Finn ES. ScienceDirect Challenges and opportunities of mesoscopic brain mapping with fMRI. COBEHA. 2021;40:189-200.
- Jia K, Kourtzi Z. Protocol A protocol for ultra-high field laminar fMRI in the human brain brain. STAR Protoc. 2021;2(2):100415.
- Shao X, Guo F, Shou Q, et al. Laminar perfusion imaging with zoomed arterial spin labeling at 7 Tesla. bioRxiv. 2021:1-20.
- Uğurbil K. Ultrahigh field and ultrahigh resolution fMRI. Curr Opin Biomed Eng. 2021;18.
- Iamshchinina P, Kaiser D, Yakupov R, et al. Perceived and mentally rotated contents are differentially represented in cortical layers of V1. bioRxiv. 2021;20(11):766.
- Wang F, Dong Z, Wald LL, Polimeni JR, Setsompop K. Simultaneous pure T2 and varying T2′-weighted BOLD fMRI using Echo Planar Time-resolved Imaging (EPTI) for mapping laminar fMRI responses. Neuroimage. 2021:1-24.
- Yun SD, Pais-roldán P, Palomero-gallagher N, Shah NJ. Mapping of Whole-Brain Resting-State Networks with Half-Millimetre. HBM. 2022.
- Fracasso A, Dumoulin SO, Petridou N. Point-spread function of the BOLD response across columns and cortical depth in human extra-striate cortex. Prog Neurobiol. 2021:104947.
- Scheeringa R, Bonnefond M, van Mourik T, Jensen O, Norris DG, Koopmans PJ. Relating neural oscillations to laminar fMRI connectivity. bioRxiv. 2020.
- Scheffler K, Engelmann J, Heule R. BOLD sensitivity and vessel size specificity along CPMG and GRASE echo trains. Magn Reson Imaging. 2021:1-8.
- Uludag K, Havlicek M. Determining laminar neuronal activity from BOLD fMRI using a generative model. Prog Neurobiol. 2021;(April):102055.
- Mourik T Van, Koopmans PJ, Bains LJ, Norris DG, Fm J. Investigation of layer specific BOLD during visual attention in the human visual cortex. bioRxiv. 2021:1-17.
- Zaretskaya N. Zooming-in on higher-level vision: High-resolution fMRI for understanding visual perception and awareness. Prog Neurobiol. 2021;(November 2020):101998.
- Taso M, Munsch F, Zhao L, Alsop DC. Regional and depth-dependence of cortical blood-flow assessed with high-resolution Arterial Spin Labeling (ASL). J Cereb Blood Flow Metab. 2021.
- Stanley OW, Kuurstra AB, Klassen LM, Menon RS, Gati JS. Effects of phase regression on high-resolution functional MRI of the primary visual cortex. Neuroimage. 2021;227(December 2020):117631.
- Park S, Torrisi S, Townsend JD, Beckett A, Feinberg DA. Highly accelerated submillimeter resolution 3D GRASE with controlled T2 blurring in T2-weighted functional MRI at 7 Tesla: A feasibility study. Magn Reson Med. 2021;85(5):2490-2506.
- van Dijk JA, Fracasso A, Petridou N, Dumoulin SO. Validating Linear Systems Analysis for Laminar fMRI: Temporal Additivity for Stimulus Duration Manipulations. Brain Topogr. 2021;34(1):88-101.
- Markuerkiaga I, Marques JP, Gallagher TE, Norris DG. Estimation of Laminar BOLD Activation Profiles using Deconvolution with a Physiological Point Spread Function. Journal of Neuroscience Methods. 2021:1-28.
- Schreiber S, Northall A, Weber M, et al. Topographical layer imaging as a tool to track neurodegenerative disease spread in M1. Nat Rev Neurosci. 2021;22(1):69.
- Huang P, Correia MM, Rua C, Rodgers CT, Henson N, Carlin JD. Correcting for Superficial Bias in 7T Gradient Echo fMRI. Frontiers in Neuroscience. 2021.
2020
- Pais-Roldán P, Yun SD, Palomero-Gallagher N, Shah NJ. Cortical depth-dependent human fMRI of resting-state networks using EPIK. bioRxiv. 2020:1-26.
- Weldon KB, Olman CA. Forging a path to mesoscopic imaging success with ultra-high field functional magnetic resonance imaging. Philos Trans B. 2020.
- Zamboni E, Kemper VG, Goncalves NR, et al. Fine-scale computations for adaptive processing in the human brain. Elife. 2020;9:1-21.
- Navarro KT, Sanchez MJ, Engel SA, Olman CA, Weldon KB. Depth-dependent functional MRI responses to chromatic and achromatic stimuli throughout V1 and V2. Neuroimage. 2020:117520.
- Bollmann S, Barth M. New acquisition techniques and their prospects for the achievable resolution of fMRI. Prog Neurobiol. 2020:ahead of print.
- Báez-Yánez MG, Siero JC, Petridou N. A statistical 3D model of the human cortical vasculature to compute the hemodynamic fingerprint of the BOLD fMRI signal. bioRxiv. 2020;31(0):1-63.
- Finn ES, Huber L, Bandettini PA. Higher and deeper: Bringing layer fMRI to association cortex. Prog Neurobiol. 2020;101930.
- Kay K, Jamison KW, Zhang RY, Uğurbil K. A temporal decomposition method for identifying venous effects in task-based fMRI. Nat Methods. 2020;17(10):1033-1039.
- Kashyap S, Ivanov D, Havlicek M, Huber L, Poser BA, Uludağ K. Sub-millimetre resolution laminar fMRI using arterial spin labelling in humans at 7T. bioRxiv. 2020:1-45.
- Kuehn E, Pleger B. Encoding schemes in somatosensation: From micro- to meta-topography. Neuroimage. 2020;223(November 2019).
- Haarsma J, Kok P, Browning M. The promise of layer-specific neuroimaging for testing predictive coding theories of psychosis. Schizophrenia Research. 2020.
- Morgan AT, Nothnagel N, Petro LS, Goense J, Muckli L. High-resolution line-scanning reveals distinct visual response properties across human cortical layers. bioRxiv. 2020:1-17.
- Zaretskaya N, Bause J, Polimeni JR, Grassi PR, Scheffler K, Bartels A. Eye-selective fMRI activity in human primary visual cortex: Comparison between 3T and 9.4T, and effects across cortical depth. Neuroimage. 2020;220.
- McColgan P, Helbling S, Vaculčiaková L, et al. Relating quantitative 7T MRI across cortical depths to cytoarchitectonics, gene expression and connectomics: A framework for tracking neurodegenerative disease. bioRxiv. 2020.
- Mccolgan P, Joubert J, Tabrizi SJ. The human motor cortex microcircuit: insights for neurodegenerative disease. Nat Rev Neurosci. 2020;3(4):1-15.
- Marquardt I, Weerd P De, Schneider M, Gulban OF, Ivanov D, Uludag K. Depth-resolved ultra-high field fMRI reveals feedback contributions to surface motion perception. 2019:1-40.
- Jia K, Zamboni E, Kemper V, et al. Recurrent Processing Drives Perceptual Plasticity. Curr Biol. 2020;30:1-11. doi:10.1016/j.cub.2020.08.016
- Hollander, Gilles et al., 2020. “Ultra-high resolution fMRI reveals origins of feedforward and feedback activity within laminae of human ocular dominance columns.” NeuroImage 2020, .
- Vizioli, Luca et al. 2020. “Multivoxel Pattern of Blood Oxygen Level Dependent Activity Can Be Sensitive to Stimulus Specific Fine Scale Responses.” Scientific Reports
- Guo F, Liu C, Qian C, et al. Layer-dependent multiplicative effects of spatial attention on contrast responses in human early visual cortex. bioRxiv. 2020.
- Zamboni E, Kemper VG, Goncalves N, et al. Suppressive recurrent and feedback computations for adaptive processing in the human brain. bioRxiv. 2020.
- Aitken F, Menelaou G, Warrington O, et al. Prior expectations evoke stimulus templates in the deep layers of V1. Plos Biology. 2020;44(0):2020.02.13.947622.
- Margalit E, Jamison KW, Weiner KS, et al. Ultra-high-resolution fMRI of human ventral temporal cortex reveals differential representation of categories and domains. J Neurosci. 2020.
- Hendriks AD, D’Agata F, Raimondo L, et al. Pushing functional MRI spatial and temporal resolution further: High-density receive arrays combined with shot-selective 2D CAIPIRINHA for 3D echo-planar imaging at 7 T. NMR Biomed. 2020.
- Persichetti AS, Avery JA, Huber L, Merriam EP, Martin A. Layer-Specific Contributions to Imagined and Executed Hand Movements in Human Primary Motor Cortex. SSRN Electron J. 2020:1-5.
- Dijk JA Van, Fracasso A, Petridou N, Dumoulin SO. Linear systems analysis for laminar fMRI : Evaluating BOLD amplitude scaling for luminance contrast manipulations. Sci Rep. 2020;10:5462.
- Huber L, Finn ES, Chai Y, et al. Layer-dependent functional connectivity methods. Prog Neurobiol. 2020
- Guo, Fanhua et al. 2020. “Layer-Dependent Multiplicative Effects of Spatial Attention on Contrast Responses in Human Early Visual Cortex.” bioRxiv: preprint.
- Guidi, M et al. 2020. “Cortical Laminar Resting-State Fluctuations Scale with the Hypercapnic Bold Response.” HBM: ahead of print.
- Huber, Laurentius et al. 2020. “Sub-Millimeter FMRI Reveals Multiple Topographical Digit Representations That Form Action Maps in Human Motor Cortex.” NeuroImage 208: 116463.
- Gau, Remi et al. 2019. “Resolving Multisensory and Attentional Influences across Cortical Depth in Sensory Cortices.” eLife: 548933.
2019
2018
2017
2016
2015
2014
2013
2012
2011
2010
2009
2007
2002
1999
High field MRI scanners worldwide
Currently there are more then 100 ultra-high field scanners installed.
An open google map of all scanners:
Bias field correction
This post shows the overview of doing bias field correction in in SPM. Doing this helps me a lot to improve the accuracy of FreeSurfer with MP2RAGE data. The members of Polimeni’s group also use it as an additional per-processing step before giving the data to Freesurfer.
The SPM bis field correction is part of the segmentation pipeline in SPM. I use the following bash script and the following matlab stript:
Bias_field_script_job.m and start_bias_field.sh.
Using a standalone Nifti I/O in C++
In this blog post, I describe how you can build your own standalone C++ program to analyze nii data without any dependencies beyond C++. Hence, it will work without the libraries from other fMRI software packages, e.g. odin, afni. The only tricky part is to use a stand alone nii I/O to write your own C++ evaluation programs. Here, I do it by incorporating the few files that i borrowed from the afni source code.
How to deal with Ghosts in high-res EPI
Background
Ghost are the biggest limitation in high res fMRI. Similar to low resolution fMRI, ghosts in sub-millimeter EPI are arising from mismatch of k-space lines. This mismatch can be associated with (A) the actual readout itself or (B) inappropriate GRAPPA auto calibration data.
Here I try to make notes of strategies that I found helpful to minimize ghosts.
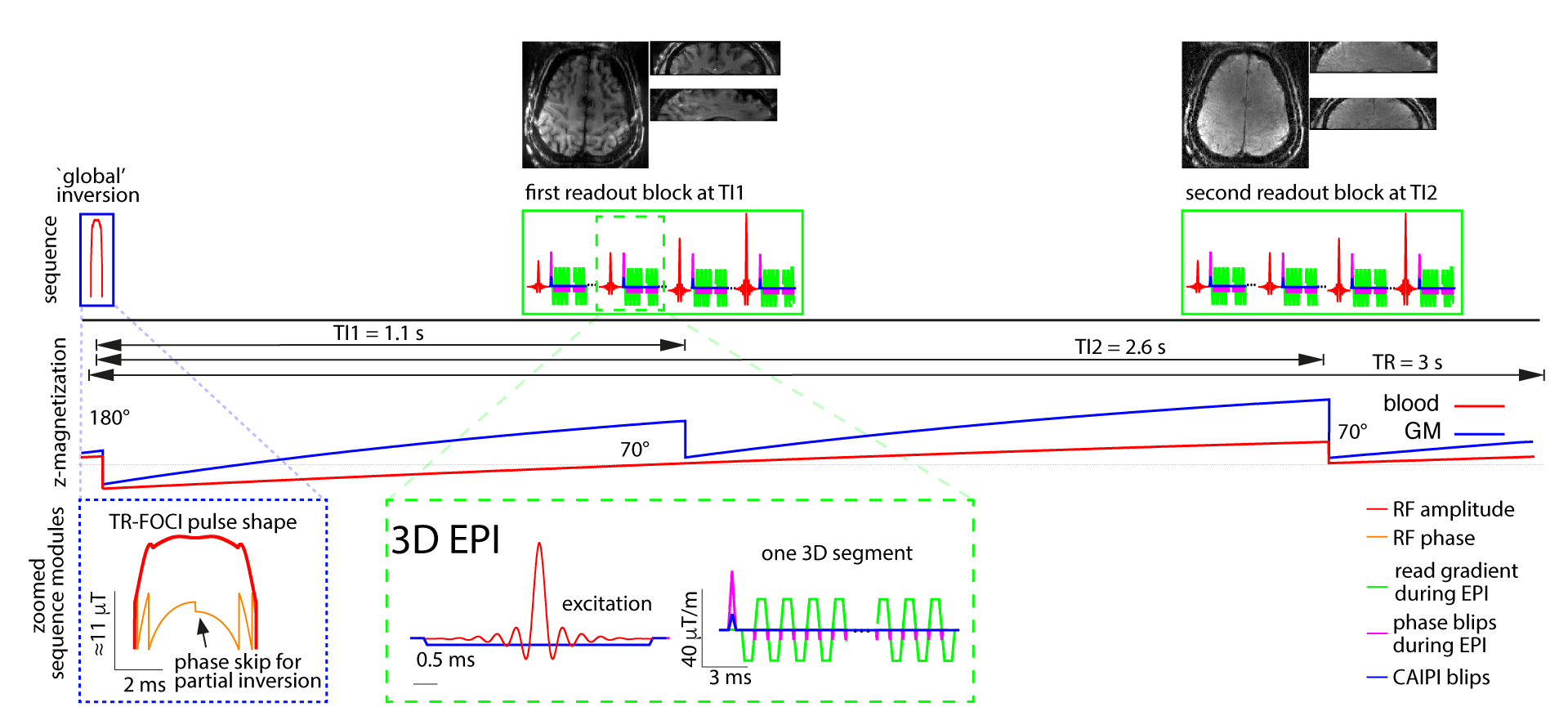
SS-SI-VASO Sequence Manual
The paper that explains the sequence and its parameter regime is can be found here: https://www.ncbi.nlm.nih.gov/pubmed/23963641. The sequence diagram looks something like this: